What Gives Elements in a Family Similar Properties
Periodic trends in backdrop such as diminutive size and ionic size, ionization free energy, electron analogousness, and electronegativity illustrate the strong connection betwixt the chemical properties and the reactivity of the elements and their positions in the periodic table. In this section, we explore that connection by focusing on 2 periodic properties that correlate strongly with the chemical behavior of the elements: valence electron configurations and Mulliken electronegativities.
The Main Grouping Elements
We have said that elements with the same valence electron configuration (i.e., elements in the same cavalcade of the periodic table) oft have similar chemical science. This correlation is particularly evident for the elements of groups one, 2, 3, 13, xvi, 17, and 18. The intervening families in the p block (groups fourteen and 15) straddle the diagonal line separating metals from nonmetals. The lightest members of these ii families are nonmetals, so they react differently compared to the heaviest members, which are metals. We begin our survey with the brine metals (grouping 1), which incorporate simply a single electron outside a noble gas electron configuration, and cease with the noble gases (group 18), which have total valence electron shells.
Group 1: The Alkali Metals
The elements of group 1 are chosen the alkali metals. Alkali (from the Arabic al-qili, significant "ashes of the saltwort plant from salt marshes") was a general term for substances derived from forest ashes, all of which possessed a bitter sense of taste and were able to neutralize acids. Although oxides of both grouping 1 and group 2 elements were obtained from wood ashes, the alkali metals had lower melting points.
Potassium and sodium were first isolated in 1807 by the British pharmacist Sir Humphry Davy (1778–1829) by passing an electric electric current through molten samples of potash (KiiCOthree) and soda ash (NaiiCO3). The potassium flare-up into flames equally soon as it was produced considering information technology reacts readily with oxygen at the college temperature. All the same, the group 1 elements, like the group 2 elements, become less reactive with air or h2o every bit their atomic number decreases. The heaviest chemical element (francium) was not discovered until 1939. Information technology is so radioactive that studying its chemical science is very difficult.
The brine metals have ns 1 valence electron configurations and the lowest electronegativity of whatsoever group; hence they are frequently referred to as being electropositive elements. Equally a outcome, they have a strong tendency to lose their single valence electron to form compounds in the +1 oxidation country, producing the EX monohalides and the EiiO oxides.
Because they are so reactive, pure grouping 1 elements are powerful reducing agents that are used in lithium batteries and cardiac pacemakers. Sodium salts such as common table salt (NaCl), blistering soda (NaHCO3), soda ash (Na2CO3), and caustic soda (NaOH) are important industrial chemicals. Other compounds of the brine metals are of import in biology. For example, because potassium is required for establish growth, its compounds are used in fertilizers, and lithium salts are used to care for manic-depressive, or bipolar, disorders.

Potassium burning. A piece of potassium dropped in a chalice of water will burn equally it skips across the top of the water.
Group 2: The Alkali metal Earth Metals
The elements of grouping two are collectively referred to as the alkaline earth metals, a name that originated in the Heart Ages, when an "earth" was defined as a substance that did not melt and was not transformed by fire. Alkalis that did non melt easily were called "alkaline metal earths."
Recall that the trend in most groups is for the lightest member to have properties that are quite dissimilar from those of the heavier members. Consistent with this trend, the properties of the lightest chemical element—in this case, beryllium—tend to be different from those of its heavier congeners, the other members of the grouping. Glucinium is relatively unreactive simply forms many covalent compounds, whereas the other group members are much more reactive metals and form ionic compounds. As is the case with the brine metals, the heaviest chemical element, radium, is highly radioactive, making its size difficult to measure. Radium was discovered in 1902 by Marie Curie (1867–1934; Nobel Prize in Chemistry 1903 and Nobel Prize in Chemistry 1911), who, with her husband, Pierre, isolated 120 mg of radium chloride from tons of residues from uranium mining. (For more than information about radioactive decay, come across Chapter 1 "Introduction to Chemical science", Department i.5 "The Atom".)
All the alkaline earth metals accept ns 2 valence electron configurations, and all take electronegativities less than 1.half-dozen. This means that they bear chemically equally metals (although beryllium compounds are covalent) and lose the two valence electrons to form compounds in the +ii oxidation state. Examples include the dihalides (EX2) and the oxides (EO).
Compounds of the group two elements have been commercially of import since Egyptian and Roman times, when blocks of limestone or marble, which are both CaCO3, were used as building materials, and gypsum (CaSO4·2 H2O) or lime (CaO) was used equally mortar. Calcium sulfate is still used in Portland cement and plaster of Paris. Magnesium and glucinium form lightweight, high-strength alloys that are used in the aerospace, automotive, and other loftier-tech industries. As you learned in Chapter 6 "The Construction of Atoms", ane of the most impressive uses of these elements is in fireworks; strontium and barium salts, for example, give red or green colors, respectively. Except for beryllium, which is highly toxic, the grouping 2 elements are also important biologically. Bone is largely hydroxyapatite [Cav(POfour)3OH], clam shells are calcium carbonate, magnesium is part of the chlorophyll molecule in greenish plants, and calcium is important in hormonal and nerve betoken transmission. Because BaSO4 is so insoluble, it is used in "barium milk shakes" to obtain ten-rays of the gastrointestinal tract.
Grouping 13

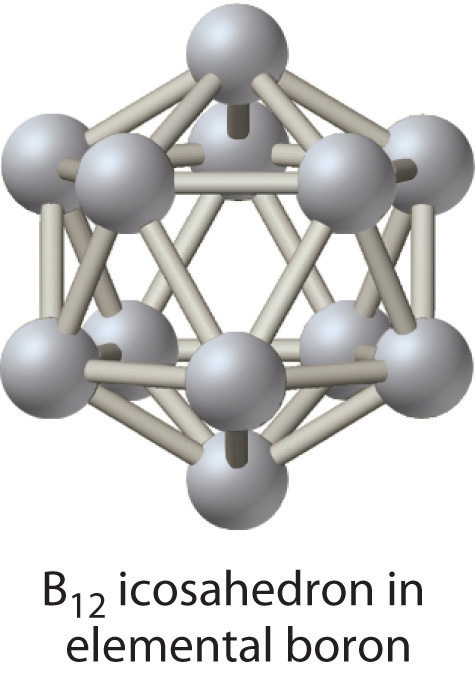
Of the group 13 elements, only the lightest, boron, lies on the diagonal line that separates nonmetals and metals. Thus boron is a semimetal, whereas the rest of the group 13 elements are metals. Elemental boron has an unusual structure consisting of B12 icosahedra covalently bonded to one another; the other elements are typical metallic solids.
No group 13 elements were known in aboriginal times, not because they are scarce—Al is the third most abundant element in Earth's chaff—but because they are highly reactive and form extremely stable compounds with oxygen. To isolate the pure elements, potent reducing agents and careful treatment were needed.
The elements of group xiii take ns 2 np 1 valence electron configurations. Consequently, two oxidation states are important: +3, from losing iii valence electrons to requite the closed-shell electron configuration of the preceding noble gas; and +1, from losing the single electron in the np subshell. Considering these elements have modest, negative electron affinities (boron'southward is just −27.0 kJ/mol), they are unlikely to acquire v electrons to reach the adjacent noble gas configuration. In fact, the chemistry of these elements is nearly exclusively characterized by +iii. Only the heaviest element (Tl) has extensive chemistry in the +1 oxidation land. Information technology loses the unmarried vip electron to produce TlX monohalides and the oxide Tl2O.
In the 19th century, aluminum was considered a precious metal. In fact, it was considered then precious that aluminum knives and forks were reserved for the French Emperor Louis Napoleon III, while his less of import guests had to be content with gold or silver cutlery. Considering of the metal'due south rarity the dedication of the Washington Monument in 1885 was celebrated past placing a 100 oz chunk of pure aluminum at the top. In dissimilarity, today aluminum is used on an enormous calibration in shipping, machine engines, armor, cookware, and beverage containers. It is valued for its combination of low density, high strength, and corrosion resistance. Aluminum is also establish in compounds that are the active ingredients in almost antiperspirant deodorants.
Compounds of boron, such equally ane class of BN, are hard, have a high melting signal, and are resistant to corrosion. They are particularly useful in materials that are exposed to extreme weather, such as aircraft turbines, brake linings, and polishing compounds. Boron is too a major component of many kinds of glasses, and sodium perborate [NatwoB2O4(OH)4] is the active ingredient in many so-called color-safe laundry bleaches.
Gallium, indium, and thallium are less widely used, but gallium arsenide is the cherry-red light-emitting diode (LED) in digital readouts in electronics, and MgGatwoOfour produces the dark-green calorie-free emitted in many xerographic machines. Compounds of thallium(I) are extremely toxic. Although Tl2SO4 is an fantabulous rat or ant poisonous substance, it is so toxic to humans that it is no longer used for this purpose.
Group 14

The group 14 elements straddle the diagonal line that divides nonmetals from metals. Of the elements in this group, carbon is a nonmetal, silicon and germanium are semimetals, and tin and atomic number 82 are metals. Equally a effect of this diversity, the structures of the pure elements vary greatly.
The ns ii np 2 valence electron configurations of group 14 gives rise to iii oxidation states: −4, in which four electrons are added to achieve the closed-beat electron configuration of the side by side noble gas; +iv, in which all iv valence electrons are lost to requite the airtight-trounce electron configuration of the preceding noble gas; and +2, in which the loss of two np two electrons gives a filled ns ii subshell.
The electronegativity of carbon is only ii.5, placing it in the middle of the electronegativity range, so carbon forms covalent compounds with a broad variety of elements and is the footing of all organic compounds. All of the grouping 14 elements form compounds in the +4 oxidation state, so all of them are able to form dioxides (from CO2 to PbO2) and tetrachlorides (CClfour and PbCliv). Only the two metallic elements, Sn and Pb, form an all-encompassing serial of compounds in the +2 oxidation state. Tin salts are sprayed onto glass to make an electrically conductive coating, and then the drinking glass is used in the industry of frost-costless windshields. Lead sulfate is formed when your car battery discharges.
Carbon has at to the lowest degree 4 allotropes (forms or crystal structures) that are stable at room temperature: graphite; diamond; a group of related muzzle structures called fullerenesOne of at least four allotropes of carbon comprising a group of related cage structures. (such as C60); and nanotubesI of at least four allotropes of carbon that are cylinders of carbon atoms and are intermediate in construction between graphite and the fullerenes. , which are cylinders of carbon atoms (Figure seven.18 "Four Allotropes of Carbon"). Graphite consists of extended planes of covalently bonded hexagonal rings. Because the planes are not linked by covalent bonds, they tin slide beyond i some other easily. This makes graphite ideally suited equally a lubricant and as the "atomic number 82" in pb pencils. Graphite too provides the black color in inks and tires, and graphite fibers are used in high-tech items such as golf clubs, tennis rackets, airplanes, and sailboats because of their lightweight, forcefulness, and stiffness.
Figure 7.18 Iv Allotropes of Carbon
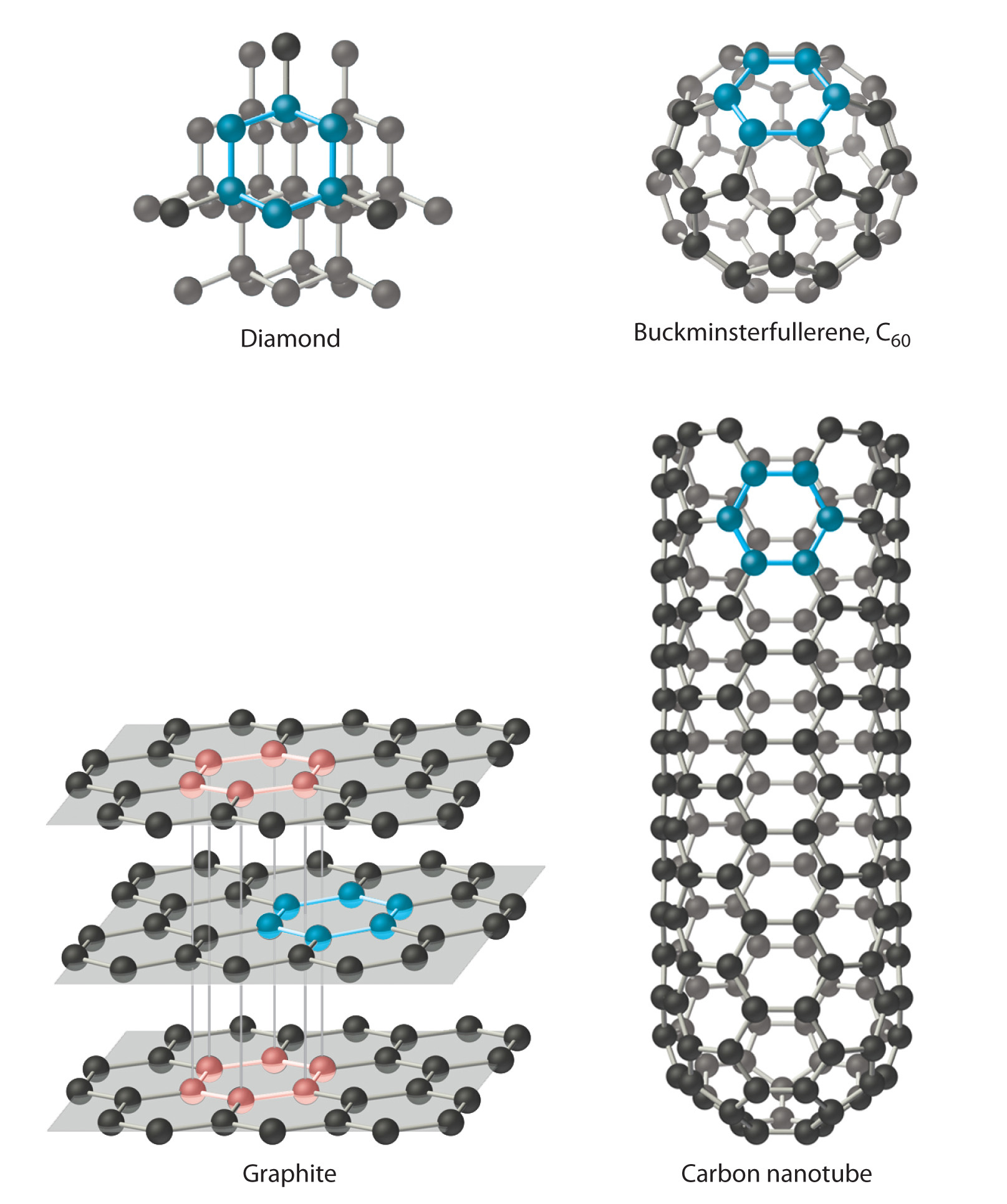
Diamond consists of a rigid three-dimensional array of carbon atoms, making information technology one of the hardest substances known. In contrast, graphite forms from extended planes of covalently bonded hexagonal rings of carbon atoms that can slide across ane some other easily. Fullerenes are spherical or ellipsoidal molecules with half dozen- and five-membered rings of carbon atoms, and nanotubes are sheets of graphite rolled upwards into a cylinder.
In contrast to the layered construction of graphite, each carbon atom in diamond is bonded to 4 others to form a rigid three-dimensional array, making diamond ane of the hardest substances known; consequently, it is used in manufacture as a cutting tool. Fullerenes, on the other manus, are spherical or ellipsoidal molecules with six- and five-membered rings of carbon atoms; they are volatile substances that dissolve in organic solvents. Fullerenes of extraterrestrial origin have been constitute in meteorites and take been discovered in a cloud of cosmic grit surrounding a afar star, which makes them the largest molecules ever seen in space. Carbon nanotubes, intermediate in construction betwixt graphite and the fullerenes, can exist described as sheets of graphite that have been rolled up into a cylinder or, alternatively, fullerene cages that have been stretched in one direction. Carbon nanotubes are being studied for use in the construction of molecular electronic devices and computers. For example, fabrics that are dipped in an ink of nanotubes and then pressed to thin out the coating are turned into batteries that maintain their flexibility. This creates "wearable electronics" and allows for the possibility of incorporating electronics into flexible surfaces. When applied to a t-shirt, for example, the t-shirt is converted into an "due east-shirt."
Silicon is the second must abundant element in Earth's crust. Both silicon and germanium take strong, 3-dimensional network structures similar to that of diamond. Sand is primarily SiOii, which is used commercially to make glass and prevent caking in food products. Complex compounds of silicon and oxygen with elements such every bit aluminum are used in detergents and talcum powder and as industrial catalysts. Because silicon-bit technology laid the foundation for the modern electronics industry, the San Jose region of California, where many of the about important advances in electronics and computers were developed, has been nicknamed "Silicon Valley."
Elemental can and pb are metal solids. Tin is primarily used to brand alloys such as bronze, which consists of tin and copper; solder, which is tin can and lead; and pewter, which is tin can, antimony, and copper.
In ancient times, pb was used for everything from pipes to cooking pots because it is hands hammered into different shapes. In fact, the term plumbing is derived from plumbum, the Latin name for lead. Lead compounds were used equally pigments in paints, and tetraethyllead was an important antiknock agent in gasoline. Now, nonetheless, pb has been banned from many uses because of its toxicity, although it is even so widely used in pb storage batteries for automobiles. In previous centuries, pb salts were frequently used equally medicines. Bear witness suggests, for case, that Beethoven's death was acquired by the application of various atomic number 82-containing medicines by his physician. Beethoven contracted pneumonia and was treated with atomic number 82 salts, merely in addition, he suffered from a serious liver ailment. His physician treated the ailment by repeatedly puncturing his abdominal cavity and then sealing the wound with a lead-laced poultice. It seems that the repeated doses of lead compounds contributed to Beethoven'southward death.
Group fifteen: The Pnicogens

The group fifteen elements are called the pnicogensThe elements in group 15 of the periodic tabular array. —from the Greek pnigein, pregnant "to choke," and genes, meaning "producing"—ostensibly because of the noxious fumes that many nitrogen and phosphorus compounds produce. This family has five stable elements; 1 isotope of bismuth (209Bi) is nonradioactive and is the heaviest nonradioactive isotope of any element. In one case again, the lightest member of the family has unique properties. Although both nitrogen and phosphorus are nonmetals, nitrogen under standard conditions is a diatomic gas (Due north2), whereas phosphorus consists of iii allotropes: white, a volatile, low-melting solid consisting of P4 tetrahedra; a red solid comprised of Pviii, P9, and P10 cages linked past P2 units; and black layers of corrugated phosphorus sheets. The adjacent two elements, arsenic and antimony, are semimetals with extended 3-dimensional network structures, and bismuth is a silvery metal with a pink tint.
All of the pnicogens have ns 2 np 3 valence electron configurations, leading to three common oxidation states: −3, in which three electrons are added to give the closed-shell electron configuration of the side by side noble gas; +5, in which all five valence electrons are lost to give the airtight-trounce electron configuration of the preceding noble gas; and +3, in which only the three np electrons are lost to give a filled ns 2 subshell. Considering the electronegativity of nitrogen is like to that of chlorine, nitrogen accepts electrons from most elements to grade compounds in the −3 oxidation land (such every bit in NH3). Nitrogen has only positive oxidation states when combined with highly electronegative elements, such as oxygen and the halogens (e.g., HNOthree, NF3). Although phosphorus and arsenic can combine with agile metals and hydrogen to produce compounds in which they have a −three oxidation land (PH3, for case), they typically reach oxidation states of +iii and +5 when combined with more electronegative elements, such every bit PCl3 and H3POfour. Antimony and bismuth are relatively unreactive metals, but form compounds with oxygen and the halogens in which their oxidation states are +3 and +5 (equally in Bi2O3 and SbF5).
Although it is present in most biological molecules, nitrogen was the last pnicogen to be discovered. Nitrogen compounds such as ammonia, nitric acid, and their salts are used agriculturally in huge quantities; nitrates and nitrites are used every bit preservatives in meat products such as ham and bacon, and nitrogen is a component of nearly all explosives.
Phosphorus, besides, is essential for life, and phosphate salts are used in fertilizers, toothpaste, and baking pulverisation. One, phosphorus sulfide, P4Sthree, is used to ignite modern prophylactic matches. Arsenic, in contrast, is toxic; its compounds are used as pesticides and poisons. Antimony and bismuth are primarily used in metal alloys, but a bismuth chemical compound is the active ingredient in the pop antacid medication Pepto-Bismol.
Group xvi: The Chalcogens

The group sixteen elements are often referred to as the chalcogensThe elements in group 16 of the periodic table. —from the Greek chalk, meaning "copper," and genes, significant "producing"—considering the most ancient copper ore, copper sulfide, is also rich in two other group 16 elements: selenium and tellurium. Once once more, the lightest fellow member of the family has unique properties. In its most common pure grade, oxygen is a diatomic gas (O2), whereas sulfur is a volatile solid with S8 rings, selenium and tellurium are gray or silver solids that have bondage of atoms, and polonium is a argent metallic with a regular array of atoms. Like astatine and radon, polonium is a highly radioactive metallic element.
All of the chalcogens have ns 2 np 4 valence electron configurations. Their chemical science is dominated past three oxidation states: −two, in which two electrons are added to attain the airtight-beat out electron configuration of the side by side element of group 0; +six, in which all half-dozen valence electrons are lost to give the airtight-beat electron configuration of the preceding noble gas; and +4, in which only the four np electrons are lost to give a filled ns 2 subshell. Oxygen has the second highest electronegativity of whatsoever element; its chemistry is dominated by the −2 oxidation state (every bit in MgO and HiiO). No compounds of oxygen in the +4 or +6 oxidation country are known. In contrast, sulfur can form compounds in all three oxidation states. Sulfur accepts electrons from less electronegative elements to requite H2S and NaiiS, for example, and it donates electrons to more electronegative elements to give compounds such as And thenii, SO3, and SF6. Selenium and tellurium, near the diagonal line in the periodic table, acquit similarly to sulfur merely are somewhat more likely to exist found in positive oxidation states.
Oxygen, the 2d virtually electronegative chemical element in the periodic table, was not discovered until the late 18th century, even though it constitutes 20% of the atmosphere and is the most abundant element in Globe's crust. Oxygen is essential for life; our metabolism is based on the oxidation of organic compounds by O2 to produce CO2 and H2O. Commercially, oxygen is used in the conversion of pig iron to steel, as the oxidant in oxyacetylene torches for cutting steel, as a fuel for the US space shuttle, and in hospital respirators.
Sulfur is the brimstone in "burn down and brimstone" from aboriginal times. Partly as a result of its long history, information technology is employed in a wide variety of commercial products and processes. In fact, as you lot learned in Chapter 2 "Molecules, Ions, and Chemical Formulas", more than sulfuric acrid is produced worldwide than whatsoever other chemical compound. Sulfur is used to cantankerous-link the polymers in safe in a process called vulcanization, which was discovered by Charles Goodyear in the 1830s and commercialized by Benjamin Goodrich in the 1870s. Vulcanization gives rubber its unique combination of strength, elasticity, and stability.
Selenium, the just other commercially important chalcogen, was discovered in 1817, and today it is widely used in light-sensitive applications. For example, photocopying, or xerography, from the Greek xèrós, meaning "dry," and graphia, meaning "writing," uses selenium films to transfer an image from one piece of paper to another, while compounds such as cadmium selenide are used to measure light in photographic light meters and automatic streetlights.
Group 17: The Halogens

The term halogen, derived from the Greek háls, meaning "common salt," and genes, pregnant "producing," was start applied to chlorine because of its tendency to react with metals to form salts. All of the halogens have an ns 2 np five valence electron configuration, and all but astatine are diatomic molecules in which the 2 halogen atoms share a pair of electrons. Diatomic Fii and Cl2 are pale yellowish-green and stake greenish gases, respectively, while Brii is a red liquid, and I2 is a purple solid. The halogens were not isolated until the 18th and 19th centuries.
Because of their relatively high electronegativities, the halogens are nonmetallic and generally react past gaining one electron per atom to reach a element of group 0 electron configuration and an oxidation state of −i. Halides are produced according to the following equation, in which X denotes a halogen:
Equation 7.16
2 Due east +nXii → two EX due north
If the chemical element Due east has a low electronegativity (as does Na), the product is typically an ionic halide (NaCl). If the element E is highly electronegative (equally P is), the product is typically a covalent halide (PCl5). Ionic halides tend to exist nonvolatile substances with high melting points, whereas covalent halides tend to be volatile substances with low melting points. Fluorine is the most reactive of the halogens, and iodine the least, which is consistent with their relative electronegativities (Effigy 7.15 "Pauling Electronegativity Values of the ").Every bit we shall see in subsequent chapters, all the same, factors such as bond strengths are also important in dictating the reactivities of these elements. In fact, fluorine reacts with about all elements at room temperature. Under more extreme weather, it combines with all elements except helium, neon, and argon.
The halogens react with hydrogen to form the hydrogen halides (HX):
Equation 7.17
Htwo(g) + X2(k,l,s) → 2 HX(g)
Fluorine is so reactive that any substance containing hydrogen, including coal, woods, and even water, will flare-up into flames if it comes into contact with pure F2.
Considering it is the near electronegative chemical element known, fluorine never has a positive oxidation state in whatsoever compound. In dissimilarity, the other halogens (Cl, Br, I) form compounds in which their oxidation states are +i, +3, +5, and +7, as in the oxoanions, XO northward −, where due north = 1–4. Considering oxygen has the second highest electronegativity of any element, it stabilizes the positive oxidation states of the halogens in these ions.
All of the halogens except astatine (which is radioactive) are commercially of import. NaCl in salt water is purified for utilise as table salt. Chlorine and hypochlorite (OCl−) salts are used to sanitize public water supplies, swimming pools, and wastewater, and hypochlorite salts are also used as bleaches because they oxidize colored organic molecules. Organochlorine compounds are used as drugs and pesticides. Fluoride (commonly in the class of NaF) is added to many municipal h2o supplies to assist prevent molar decay, and bromine (in AgBr) is a component of the calorie-free-sensitive blanket on photographic film. Considering iodine is essential to life—it is a primal component of the hormone produced by the thyroid gland—small amounts of KI are added to table salt to produce "iodized table salt," which prevents thyroid hormone deficiencies.
Group xviii: The Noble Gases
The noble gases are helium, neon, argon, krypton, xenon, and radon. All have filled valence electron configurations and therefore are unreactive elements found in nature as monatomic gases. The noble gases were long referred to every bit either "rare gases" or "inert gases," but they are neither rare nor inert. Argon constitutes well-nigh one% of the temper, which as well contains small amounts of the lighter group eighteen elements, and helium is found in large amounts in many natural gas deposits. The group's perceived "rarity" stems in part from the fact that the noble gases were the concluding major family of elements to exist discovered.
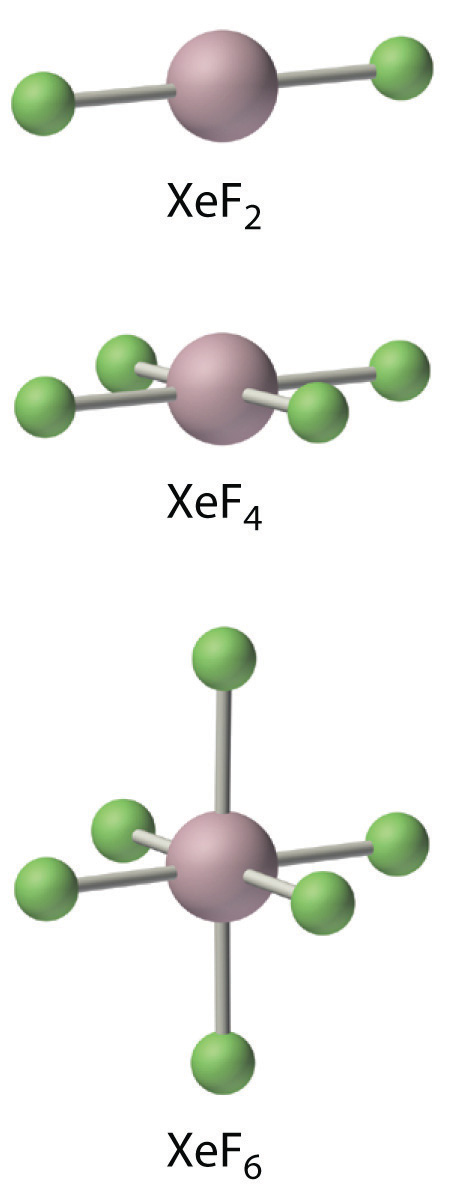
The noble gases accept EA ≥ 0, then they exercise non form compounds in which they have negative oxidation states. Because ionization energies decrease down the column, the only noble gases that class compounds in which they have positive oxidation states are Kr, Xe, and Rn. Of these three elements, only xenon forms an extensive series of compounds. The chemistry of radon is severely limited by its extreme radioactivity, and the chemistry of krypton is limited by its loftier ionization energy (1350.8 kJ/mol versus 1170.iv kJ/mol for xenon). In essentially all its compounds, xenon is bonded to highly electronegative atoms such every bit fluorine or oxygen. In fact, the only significant reaction of xenon is with elemental fluorine, which can give XeF2, XeF4, or XeFhalf dozen. Oxides such as XeO3 are produced when xenon fluorides react with water, and oxidation with ozone produces the perxenate ion [XeOsix four−], in which xenon acquires a +viii oxidation state past formally altruistic all 8 of its valence electrons to the more electronegative oxygen atoms. In all of its stable compounds, xenon has a positive, fifty-fifty-numbered oxidation country: +2, +4, +6, or +8. The actual stability of these compounds varies greatly. For case, XeO3 is a stupor-sensitive, white crystalline solid with explosive ability comparable to that of TNT (trinitrotoluene), whereas another compound, Na2XeF8, is stable up to 300°C.
Although none of the element of group 0 compounds is commercially significant, the elements themselves have important applications. For example, argon is used in incandescent light bulbs, where it provides an inert atmosphere that protects the tungsten filament from oxidation, and in compact fluorescent light bulbs (CFLs). It is likewise used in arc welding and in the industry of reactive elements, such as titanium, or of ultrapure products, such as the silicon used by the electronics industry. Helium, with a boiling betoken of only 4.2 K, is used as a liquid for studying the properties of substances at very depression temperatures. It is also combined in an 80:xx mixture with oxygen used by scuba defined, rather than compressed air, when they descend to slap-up depths. Because helium is less soluble in h2o than Ntwo—a component of compressed air—replacing Northii with He prevents the germination of bubbles in blood vessels, a condition called "the bends" that can occur during rapid ascents. Neon is familiar to all of us every bit the gas responsible for the red glow in neon lights.
The Transition Metals, the Lanthanides, and the Actinides
As expected for elements with the aforementioned valence electron configuration, the elements in each cavalcade of the d cake have vertical similarities in chemic behavior. In contrast to the s- and p-block elements, however, elements in the d block also display stiff horizontal similarities. The horizontal trends compete with the vertical trends. In farther contrast to the p-block elements, which tend to have stable oxidation states that are separated past ii electrons, the transition metalsWhatsoever element in groups 3–12 in the periodic table. All of the transition elements are metals. take multiple oxidation states that are separated past simply ane electron.
Note the Pattern
The p-block elements form stable compounds in oxidation states that tend to exist separated past two electrons, whereas the transition metals have multiple oxidation states that are separated by one electron.
The grouping 6 elements, chromium, molybdenum, and tungsten, illustrate the competition that occurs between these horizontal and vertical trends. For example, the maximum oxidation state for all elements in grouping 6 is +6, achieved past losing all six valence electrons (recall that Cr has a 4s 13d 5 valence electron configuration), yet almost all the elements in the first row of the transition metals, including chromium, form compounds with the dication Yard2+, and many too form the trication M3+. As a issue, the transition metals in grouping half dozen have very different tendencies to achieve their maximum oxidation country. The most common oxidation state for chromium is +3, whereas the virtually common oxidation state for molybdenum and tungsten is +6.
Note the Blueprint
The d-cake elements brandish both strong vertical and horizontal similarities.
Groups 3 (scandium, lanthanum, actinium), eleven (copper, silvery, aureate), and 12 (zinc, cadmium, mercury) are the only transition element groups in which the oxidation land predicted by the valence electron configuration dominates the chemistry of the group. The elements of group 3 have three valence electrons outside an inner closed shell, so their chemistry is well-nigh exclusively that of the Yard3+ ions produced by losing all three valence electrons. The elements of group 11 have 11 valence electrons in an ns 1(n − i)d 10 valence electron configuration, and and so all three lose a single electron to grade the monocation One thousand+ with a closed (n − 1)d 10 electron configuration. Consequently, compounds of Cu+, Ag+, and Au+ are very common, although there is also a great deal of chemistry involving Cu2+. Similarly, the elements of group 12 all have an ns ii(n − 1)d 10 valence electron configuration, so they lose two electrons to form Thou2+ ions with an (due north − ane)d 10 electron configuration; indeed, the most important ions for these elements are Zn2+, Cd2+, and Hg2+. Mercury, withal, also forms the dimeric mercurous ion (Hg2 2+) because of a subtle residual between the energies needed to remove additional electrons and the free energy released when bonds are formed. The +three oxidation state is the most important for the lanthanidesAny of the 14 elements between (cerium) and (lutetium). and for near of the actinidesWhatsoever of the xiv elements between (thorium) and (lawrencium). .
Example 8
Based on the following information, determine the about likely identities for elements D and E.
- Element D is a shiny greyness solid that conducts electricity only moderately; it forms two oxides (DO2 and Exerciseiii).
- Chemical element Eastward is a cherry metal substance that is an excellent usher of electricity; it forms two oxides (EO and EiiO) and 2 chlorides (ECl and ECl2).
Given: physical and chemic backdrop of 2 elements
Asked for: identities
Strategy:
A Based on the conductivity of the elements, determine whether each is a metal, a nonmetal, or a semimetal. Confirm your prediction from its physical advent.
B From the compounds each element forms, make up one's mind its mutual oxidation states.
C If the chemical element is a nonmetal, it must be located in the p block of the periodic table. If a semimetal, it must lie along the diagonal line of semimetals from B to At. Transition metals can accept two oxidation states separated past 1 electron.
D From your classification, the oxidation states of the element, and its physical appearance, deduce its identity.
Solution:
- A The moderate electrical electrical conductivity of element D tells us that information technology is a semimetal. It must prevarication in the p cake of the periodic tabular array because all of the semimetals are located there. B The stoichiometry of the oxides tells u.s.a. that 2 common oxidation states for D are +4 and +6. C Element D must be located in group 16 considering the common oxidation states for the chalcogens (group xvi) include +6 (by losing all half-dozen valence elections) and +4 (by losing the iv electrons from the p subshell). Thus D is likely to be Se or Te. D Additional information is needed to distinguish between the two.
- A Element E is an excellent electrical conductor, so information technology is a metal. B The stoichiometry of the oxides and chlorides, however, tells us that common oxidation states for Eastward are +2 and +one. C Metals that can have two oxidation states separated past one electron are normally transition metals. The +ane oxidation state is characteristic of only one grouping: grouping eleven. Inside group 11, copper is the only element with common oxidation states of +1 and +ii. D Copper likewise has a blood-red hue. Thus chemical element E is probably copper.
Do
Based on the following information, decide the near probable identities for elements Thousand and J.
- Element Yard is a red liquid that does not conduct electricity. It forms 3 compounds with fluorine (GF, GF3, and GF5) and one with sodium (NaG).
- Chemical element J is a soft, deadening gray solid that conducts electricity well and forms ii oxides (JO and JOtwo).
Answer:
- Br
- Sn or Pb
Summary
The chemical families consist of elements that take the aforementioned valence electron configuration and tend to have like chemical science. The alkali metals (group 1) have ns 1 valence electron configurations and form M+ ions, while the alkaline world metals (grouping 2) have ns 2 valence electron configurations and form M2+ ions. Group 13 elements have ns 2 np 1 valence electron configurations and have an overwhelming trend to course compounds in the +three oxidation land. Elements in group 14 have ns 2 np ii valence electron configurations merely exhibit a variety of chemical behaviors because they range from a nonmetal (carbon) to metals (tin/lead). Carbon, the basis of organic compounds, has at least iv allotropes with distinct structures: diamond, graphite, fullerenes, and carbon nanotubes. The pnicogens (group 15) all accept ns two np 3 valence electron configurations; they class compounds in oxidation states ranging from −three to +5. The chalcogens (grouping xvi) have ns 2 np four valence electron configurations and react chemically by either gaining 2 electrons or by formally losing four or half-dozen electrons. The halogens (group 17) all have ns 2 np five valence electron configurations and are diatomic molecules that tend to react chemically by accepting a single electron. The noble gases (grouping 18) are monatomic gases that are chemically quite unreactive due to the presence of a filled shell of electrons. The transition metals (groups 3–10) comprise partially filled sets of d orbitals, and the lanthanides and the actinides are those groups in which f orbitals are being filled. These groups exhibit potent horizontal similarities in behavior. Many of the transition metals form M2+ ions, whereas the chemistry of the lanthanides and actinides is dominated by Yard3+ ions.
Cardinal Takeaway
- Periodic properties and the chemical beliefs of the elements correlate strongly with valence electron configurations and Mulliken electronegativities.
Conceptual Problems
-
Of the group 1 elements, which would y'all expect to be the all-time reductant? Why? Would yous await boron to be a practiced reductant? Why or why non?
-
Classify each element as a metal, a nonmetal, or a semimetal: Hf, I, Tl, S, Si, He, Ti, Li, and Sb. Which would yous expect to be adept electrical conductors? Why?
-
Classify each element equally a metallic, a nonmetal, or a semimetal: Au, Bi, P, Kr, Five, Na, and Po. Which would you expect to be good electrical insulators? Why?
-
Of the elements Kr, Xe, and Ar, why does merely xenon course an extensive series of compounds? Would you expect Xe2+ to exist a good oxidant? Why or why not?
-
Place each statement about the halogens as either truthful or false and explain your reasoning.
- Halogens have filled valence electron configurations.
- Halogens tend to form salts with metals.
- Every bit the free elements, halogens are monatomic.
- Halogens have appreciable nonmetallic character.
- Halogens tend to accept an oxidation land of −1.
- Halogens are good reductants.
-
Nitrogen forms compounds in the +5, +4, +3, +2, and −iii oxidation states, whereas Bi forms ions but in the +5 and +3 oxidation states. Propose an explanation for the differences in behavior.
-
Of the elements Mg, Al, O, P, and Ne, which would you look to grade covalent halides? Why? How practice the melting points of covalent halides compare with those of ionic halides?
-
Of the elements Li, Ga, Equally, and Xe, would yous look to course ionic chlorides? Explain your reasoning. Which are usually more than volatile—ionic or covalent halides? Why?
-
Predict the relationship betwixt the oxidative force of the oxoanions of bromine—BrO n − (northward = 1–4)—and the number of oxygen atoms present (n). Explain your reasoning.
-
The stability of the binary hydrides of the chalcogens decreases in the order HtwoO > H2S > HtwoSe > H2Te. Why?
-
Of the elements O, Al, H, and Cl, which will form a compound with nitrogen in a positive oxidation state? Write a reasonable chemical formula for an example of a binary chemical compound with each element.
-
How practice y'all explicate the differences in chemistry observed for the group 14 elements every bit you lot go down the column? Classify each group xiv element as a metal, a nonmetal, or a semimetal. Practise you lot expect the group fourteen elements to form covalent or ionic compounds? Explain your reasoning.
-
Why is the chemistry of the grouping thirteen elements less varied than the chemical science of the grouping xv elements? Would you await the chemistry of the group thirteen elements to be more or less varied than that of the group 17 elements? Explain your reasoning.
-
If y'all needed to blueprint a substitute for BaSO4, the barium milkshake used to examine the big and minor intestine by 10-rays, would BeSO4 exist an inappropriate substitute? Explain your reasoning.
-
The alkali metals have an ns 1 valence electron configuration, and consequently they tend to lose an electron to form ions with +1 charge. Based on their valence electron configuration, what other kind of ion tin can the alkali metals form? Explain your reply.
-
Would Mo or W be the more appropriate biological substitute for Cr? Explicate your reasoning.
Answer
-
Nitrogen will have a positive oxidation state in its compounds with O and Cl, because both O and Cl are more electronegative than N. Reasonable formulas for binary compounds are: N2O5 or N2O3 and NCl3.
Numerical Bug
-
Write a balanced equation for formation of XeO3 from elemental Xe and Oii. What is the oxidation country of Xe in XeOthree? Would you wait Ar to undergo an analogous reaction? Why or why non?
-
Which of the p-block elements exhibit the greatest variation in oxidation states? Why? Based on their valence electron configurations, identify these oxidation states.
-
Based on its valence electron configuration, what are the three common oxidation states of selenium? In a binary compound, what atoms bonded to Se will stabilize the highest oxidation state? the lowest oxidation state?
-
Would yous expect sulfur to be readily oxidized past HCl? Why or why non? Would you look phosphorus to be readily oxidized by sulfur? Why or why non?
-
What are the most common oxidation states for the pnicogens? What factors determine the relative stabilities of these oxidation states for the lighter and the heavier pnicogens? What is likely to be the nigh mutual oxidation country for phosphorus and arsenic? Why?
-
Of the compounds NF3, NCl3, and NIthree, which would be the to the lowest degree stable? Explain your answer. Of the ions BrO−, ClO−, or FO−, which would be the least stable? Explain your reply.
-
In an attempt to explore the chemistry of the superheavy element ununquadium, Z = 114, you isolated two distinct salts by exhaustively oxidizing metal samples with chlorine gas. These salts are found to have the formulas MCl2 and MCl4. What would be the name of ununquadium using Mendeleev'south eka-note?
-
Would you await the compound CCl2 to exist stable? SnCl2? Why or why not?
-
A newly discovered element (Z) is a good usher of electricity and reacts only slowly with oxygen. Reaction of 1 k of Z with oxygen under three different sets of atmospheric condition gives products with masses of 1.333 1000, one.668 grand, and 1.501 g, respectively. To what family of elements does Z belong? What is the atomic mass of the chemical element?
-
An unknown element (Z) is a dull, breakable powder that reacts with oxygen at high temperatures. Reaction of 0.665 gram of Z with oxygen nether two dissimilar sets of weather forms gaseous products with masses of ane.328 1000 and 1.660 g. To which family of elements does Z belong? What is the atomic mass of the chemical element?
-
Why are the brine metals such powerful reductants? Would you expect Li to be able to reduce Htwo? Would Li reduce 5? Why or why not?
-
What do you predict to be the nigh common oxidation country for Au, Sc, Ag, and Zn? Give the valence electron configuration for each element in its most stable oxidation state.
-
Consummate the following table.
Mg C Ne Fe Br Valence Electron Configuration Common Oxidation States Oxidizing Strength -
Use the post-obit information to identify elements T, X, D, and Z. Element T reacts with oxygen to form at least iii compounds: TO, TtwoO3, and TO2. Element 10 reacts with oxygen to form XO2, but 10 is also known to grade compounds in the +two oxidation state. Element D forms DtwoO3, and chemical element Z reacts vigorously and forms Z2O. Electrical conductivity measurements showed that element X exhibited electric conductivity intermediate between metals and insulators, while elements T, D, and Z were skilful conductors of electricity. Element T is a hard, lustrous, silvery metal, element X is a blue-gray metal, element D is a light, silvery metal, and element Z is a soft, low-melting metal.
-
Predict whether Cs, F2, Al, and He will react with oxygen. If a reaction volition occur, identify the products.
-
Predict whether K, Ar, O, and Al will react with Cl2. If a reaction will occur, identify the products.
-
Use the post-obit data to identify elements X, T, and Z.
- Chemical element X is a soft, silver-white metallic that is flammable in air and reacts vigorously with h2o. Its first ionization free energy is less than 500 kJ/mol, but the 2nd ionization free energy is greater than 3000 kJ/mol.
- Element T is a gas that reacts with Fii to course a series of fluorides ranging from TF2 to TFsix. Information technology is inert to virtually other chemicals.
- Element Z is a deep red liquid that reacts with fluorine to form ZF3 and with chlorine to course ZCl and ZCl3, and with iodine to form ZI. Chemical element Z also reacts with the alkali metals and alkaline earth metals.
-
Adding a reactive metal to h2o in the presence of oxygen results in a fire. In the absence of oxygen, the addition of 551 mg of the metal to water produces 6.4 mg of hydrogen gas. Handling of 2.00 g of this metal with 6.3 g of Br2 results in the formation of 3.86 g of an ionic solid. To which chemic family unit does this element belong? What is the identity of the element? Write and remainder the chemical equation for the reaction of h2o with the metallic to form hydrogen gas.
Answers
-
2 Xe + iii O2 → two XeO3
The oxidation state of xenon in XeO3 is +6. No, Ar is much more hard to oxidize than Xe.
-
The valence electron configuration of Se is [Ar]4s 23d 104p 4. Its mutual oxidation states are: +6, due to loss of all six electrons in the 4s and fourp subshells; +four, due to loss of but the four 4p electrons; and −2, due to add-on of two electrons to give an [Ar]ivsouthward twoiiid x4p 6 electron configuration, which is isoelectronic with the following noble gas, Kr. The highest oxidation land (+6) will exist stabilized by bonds to highly electronegative atoms such equally F (SeF6) and O (SeO3), while the everyman oxidation state will exist stabilized in covalent compounds by bonds to less electronegative atoms such as H (H2Se) or C [(CH3)iiSe], or in ionic compounds with cations of electropositive metals (NaiiSe).
-
All of the pnicogens accept ns 2 np 3 valence electron configurations. The pnicogens therefore tend to form compounds in three oxidation states: +v, due to loss of all five valence electrons; +three, due to loss of the 3 np 3 electrons; and −3, due to addition of 3 electrons to give a closed shell electron configuration. Bonds to highly electronegative atoms such every bit F and O will stabilize the higher oxidation states, while bonds to less electronegative atoms such as H and C will stabilize the lowest oxidation state, as will formation of an ionic compound with the cations of electropositive metals. The most common oxidation state for phosphorus and arsenic is +five.
-
Uuq =eka-atomic number 82
-
The ratios of the masses of the element to the mass of oxygen requite empirical formulas of ZO, Z2O3, and ZO2. The loftier electrical electrical conductivity of the element immediately identifies it as a metal, and the being of 3 oxides of the element with oxidation states separated by only one electron identifies it as a transition metal. If 1 g of Z reacts with 0.33 thousand O2 to give ZO, the balanced equation for the reaction must be ii Z + Otwo → 2 ZO. Using M to correspond molar mass, the ratio of the molar masses of ZO and Z is therefore:
YardZO:MZ = (MZ + MO): ThousandZ = (MZ + 16.0): MZ = i.33:ane = i.33.
Solving for MZ gives a tooth mass of 48 g/mol and an atomic mass of 48 amu for Z, which identifies it as titanium.
-
Alkali metals are powerful reductants because they accept a strong tendency to lose their ns 1 valence electron, as reflected in their low first ionization energies and electronegativities. Lithium has a more positive electron analogousness than hydrogen and a essentially lower outset ionization energy, so we expect lithium to reduce hydrogen. Transition metals have low electron affinities and do not normally form compounds in negative oxidation states. Therefore, nosotros do not expect lithium to reduce vanadium.
-
Mg C Ne Fe Br Valence Electron Configuration 3s 2 2southward 22p two 2due south 2twop 6 ivsouth 23d 6 ivs 2ivp 5 Common Oxidation States +2 −4, +4 0 +2, +3 −1, +i, +three, +5, +vii Oxidizing Strength None Weak None None Strong -
four Cs(s) + Otwo(g) → 2 Cs2O(due south) two F2(grand) + O2(yard) → OFii(g) four Al(south) + iii O2(thousand) → 2 AliiO3(s) He + Oii(g) → no reaction
-
- sodium or potassium
- xenon
- bromine
urbinafesionfluen.blogspot.com
Source: https://saylordotorg.github.io/text_general-chemistry-principles-patterns-and-applications-v1.0/s11-04-the-chemical-families.html
Postar um comentário for "What Gives Elements in a Family Similar Properties"